HIV VARIANTS

WHAT ARE HIV VARIANTS?
HIV is one of the viruses with the highest genetic variability due to its high replication and mutation rate. The reason is that when the virus wants to duplicate its genetic material (RNA), the HIV protein responsible for doing so (called reverse transcriptase) cannot remove the nucleotides that are mistakenly incorporated into the new genetic material being generated, and those errors lead to mutations.
Some mutations make the virus non-viable and disappear, and others are maintained because they do not affect the virus's growth or because they provide benefits. Therefore, there are numerous variants of HIV, studied in the field of molecular epidemiology of HIV.
CLASSIFICATION OF HIV VARIANTS
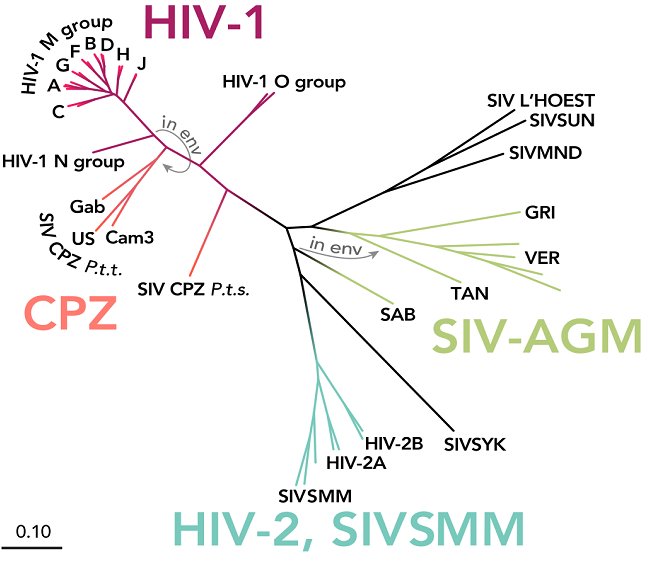
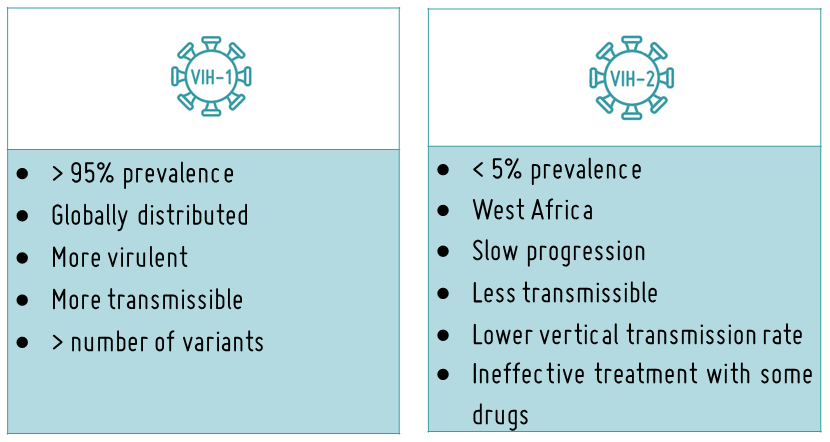
Based on its genetic homology or similarity between the composition of the viral genome (RNA), HIV is classified into two types: HIV-1 and HIV-2. HIV-1 is the most widespread and responsible for the majority of the over 38 million HIV infections worldwide. HIV-2, identified in 1986, is phylogenetically closer to the simian immunodeficiency virus (SIV) than HIV-1, being less pathogenic, less transmissible, and mainly found in certain places in West Africa.
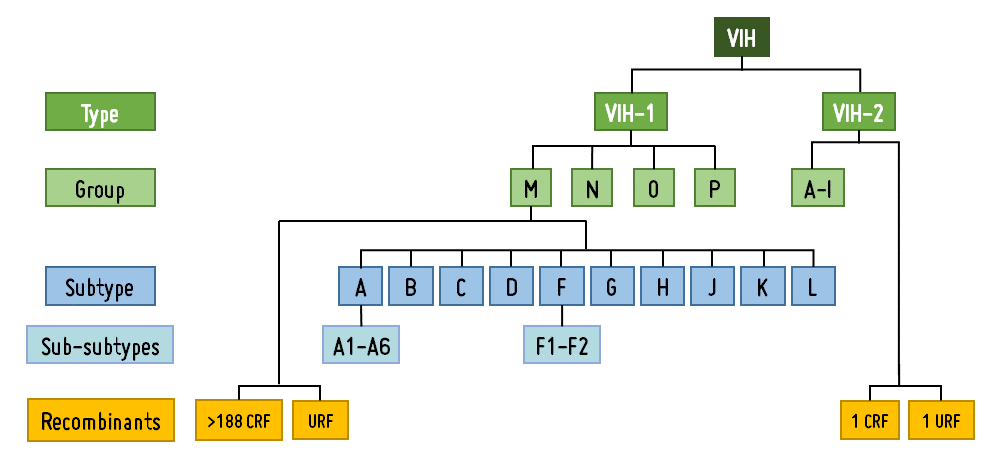
HIV-1 is classified into four major groups: group M (main), group O (outlier), group N (non-M, non-O), and group P. Groups O, N, and P have low prevalence and are restricted to West and Central sub-Saharan Africa. Group M is responsible for 97% of HIV-1 infections worldwide.
Additionally, group M viruses are subdivided into 10 subtypes (A, B, C, D, F, G, H, J, K, L) and recombinants between them (CRF, URF).
Recombinants arise through recombination following viral coinfection (successive infections) or superinfection (simultaneous infections) by two or more different subtypes and/or CRFs of the virus in the same individual. This usually occurs in places with a high infection rate and where many different variants circulate. Recombinant HIV strains can be circulating (CRF) or unique (URF).
HIV-2 has been classified into 9 groups (A-I) and 2 recombinants (CRF01_AB and URF), with groups A and B being the most prevalent.
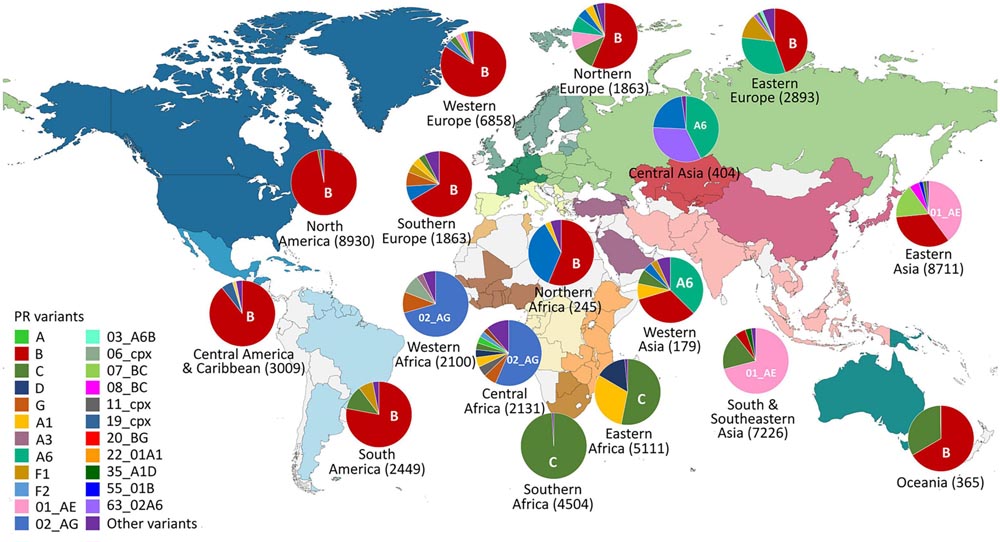
GLOBAL DISTRIBUTION OF HIV VARIANTS
The global distribution of the various variants of HIV within group M is heterogeneous. Subtype C predominates in southern Africa, subtype A in countries of the former Soviet Union, and its recombinant CRF01_AE in East and Southeast Asia. In Western Europe, America, and Australia, subtype B is predominant.
Subtype B of HIV-1 is the most studied variant and used as a model for the design and development of drugs, vaccines, and diagnostic assays, despite accounting for only 12% of HIV-1 infections worldwide. Commercial, migratory, and tourist population movements have introduced subtypes and recombinant forms of HIV-1 group M, responsible for 88% of global infections. The most prevalent HIV variant in the world is subtype C (46.6%), followed by CRF recombinants.
IMPLICATIONS OF HIV VARIANTS
The correct identification of subtypes and recombinant forms among HIV variants is highly relevant for understanding potential issues in genetic and serological diagnosis, viral load quantification, antiretroviral treatment efficacy, interpretation of the impact of drug resistance mutations (MDR), and to comprehend the disease prognosis or failures in the efficacy of vaccines under development.
The impact of HIV genetic variability on future vaccine development will be well understood if we recall the impact that new variants of SARS-CoV-2, the virus causing Covid19, had on the efficacy of some vaccines in the recent pandemic.
Certain non-B variants (variants different from subtype B) exhibit accelerated disease progression, different transmission rates, and polymorphisms or natural markers associated with ARV resistance. Therefore, the presence of non-B variants of HIV-1 not only has implications in the epidemiological field but also potentially significant clinical and biological repercussions that need to be studied.
Impact of HIV genetic variability
ONGOING PROJECTS ON HIV VARIANTS
At EPIMOLVIH, we have been working for many years with HIV variants to understand the epidemiology of the HIV pandemic and to comprehend the diagnostic, clinical, and therapeutic implications of the virus's genetic variability. We focus particularly on the identification and study of the less studied types, subtypes, and recombinants, the non-B variants, which, being less common in high-resource countries, remain the most unknown despite their high global prevalence.
After identifying these HIV variants in clinical samples from infected individuals in Spain and other parts of the world, we study their distribution and prevalence. We also detect the mutations they present, including the most frequent ones that serve as variant markers. Finally, we examine the diagnostic and therapeutic impact of this variability. Due to its importance, we include the study of variants in most of our projects. You can find our studies on HIV variants in various publications and theses. We have already studied HIV variants in Spain and in several countries in Africa and Latin America.
Do you want to collaborate with us to continue advancing in the research of HIV variants?
You too can contribute your bit to continue advancing in the research of HIV variants and understanding their presence and implications in developing countries.
Do you want to collaborate?
Join our Teaming group for just one euro per month or donate whatever you consider as an individual or company.
Together for the advancement in HIV variant research!